 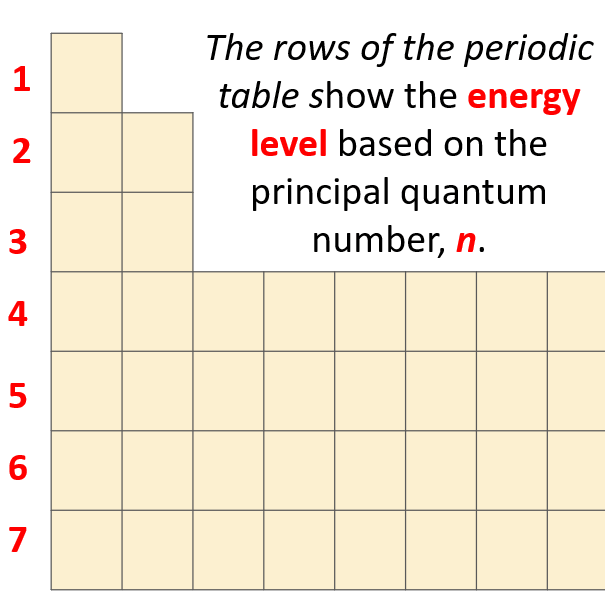 
Group 2 elements represent the second column of elements on the left side of the periodic table. Energy levels are a little like the steps of a staircase. In the periodic table, the alkaline earth metals group is found in group 2. Electrons are tiny, negatively charged particles in an atom that move around the positive nucleus at the center. The Pauli Exclusion Principle arises from more than just the electrostatic repulsion of negative electrons: it comes from fundamental physical principles that constrain all subatomic particles.\): Piece of silver. Energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. The reason that electrons tend to stay in their separate orbitals rather than piling on top of one another is the Pauli Exclusion Principle, a theorem from quantum mechanics that dictates that no two electrons can ever be in the same place. 2: Comparison of ion sizes for Groups 1, 2, 13, 16, and 17. In chemistry, periodicity refers to repeating trends in element properties on the periodic table. Usually, x-ray crystallography is employed to determine the radius for an ion. These radii will differ somewhat depending upon the technique used. This results in beautiful geometric structures called orbitals that represent the distinct regions around the nucleus that each electron traces out. The ionic radius for an atom is measured in a crystal lattice, requiring a solid form for the compound. While these electrons all stick within the atom because of their attraction to the protons, they also mutually repel each other, causing them to spread out around the nucleus in regular patterns. 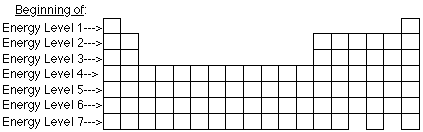
Along with the other set of 15 elements joined to them known as the lanthanides, these 15 elements. The number of elements in each row shows how many electrons it takes to fill each level. However, electrons are so small that they are. Columns are called groups, and the group number equals the number of electrons an atom of that element has in its outer shell. It is worth noting that in reality, atoms are not built by adding protons and electrons one at a time, and that this method is. The Aufbau (German for building up, construction) principle is sometimes referred to as the 'building up' principle. As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons. The Actinides are a section of 15 elements located at the extreme bottom of the periodic table. Atomic mass is the total mass of particles of matter in an atom, i.e., the masses of protons, neutrons, and electrons in an atom added together. The Aufbau principle states that an electron occupies orbitals in order from lowest energy to highest. The barium cation is written Ba 2+, not Ba +2. Note the convention of first writing the number and then the sign on a multiply charged ion. This process of filling in the electrons from the first, lowest energy level to. 3 shows how the charge on many ions can be predicted by the location of an element on the periodic table. Based on electron configurations, the periodic table can be divided into blocks denoting which sublevel is in the process of being filled. The value of the atomic radius of an element gives a measure of its atomic size. Atomic radii can be measured by measuring the distance between the nuclei of atoms in a metal. Therefore, nickel can be found in the fourth period of the periodic table. Atomic Size & Atomic Radius - Atomic Size is the distance between the center of the nucleus and the outermost shell of electrons is called the atomic radius. /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. The energy levels are built up from the level closest to the nucleus outward. The highest occupied principal energy level is the fourth, indicated by the 4 in the (4s2) portion of the configuration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
